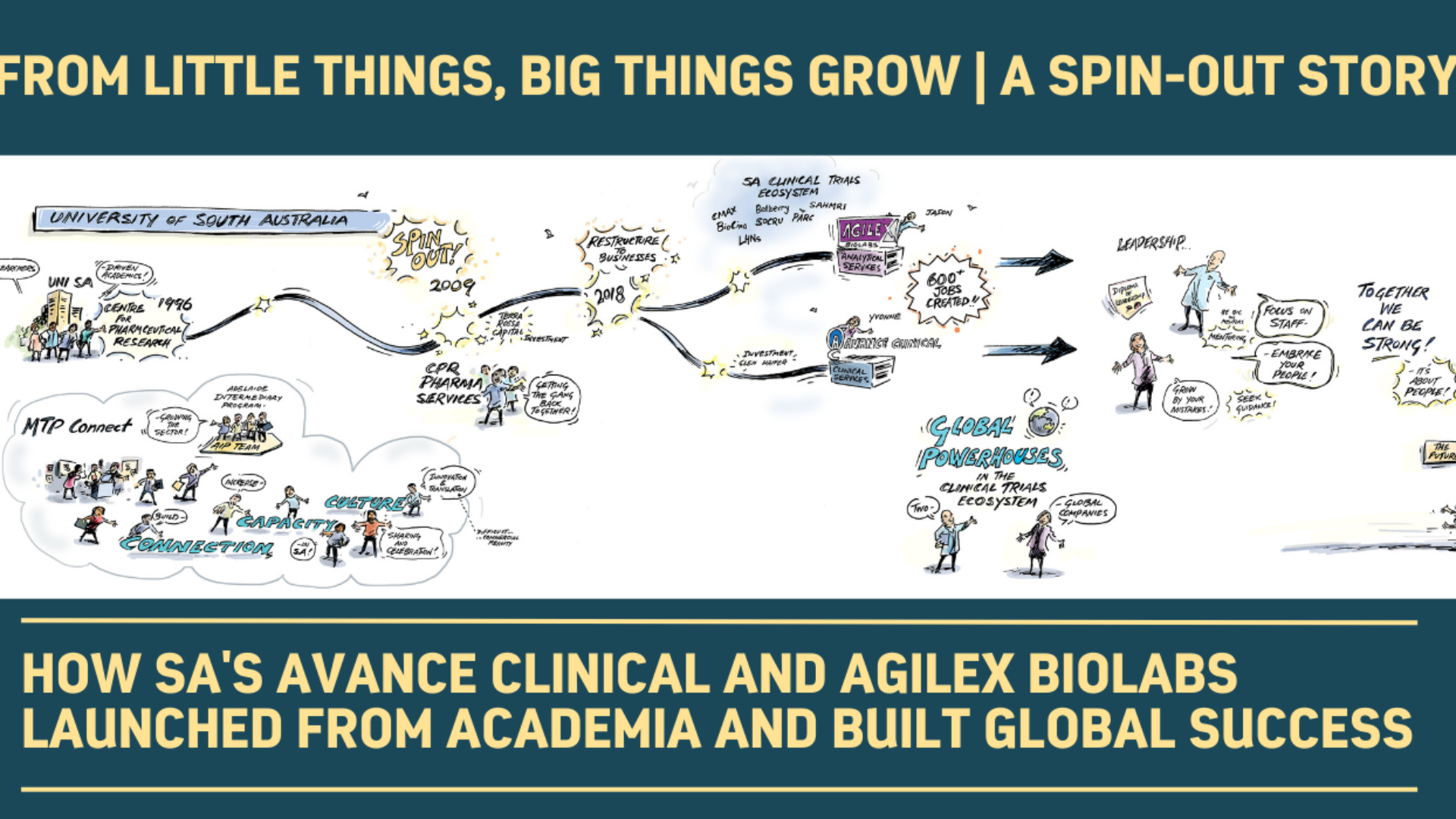
From little things, big things grow: how South Australia’s Avance Clinical and Agilex Biolabs launched from academia and built global success
Adelaide-based organisations Avance Clinical and Agilex Biolabs are prominent members of South Australia’s rapidly growing biomedical sector. From launch to present-day success, a deep dive into the two companies reveals a fascinating recent history of academic rigor, pharmaceutical analytics know-how, clinical trial expertise, entrepreneurship and customer service.
Avance Clinical CEO Yvonne Lungershausen and Agilex Biolabs COO Jason Valentine share key lessons they’ve learned throughout their respective journeys.
Avance Clinical is a leading Contract Research Organisation (CRO) providing high-quality clinical research services to international biotechs, primarily in the United States, Asia and the European Union. Agilex Biolabs is Australia’s largest bioanalytical and toxicology laboratory, supporting preclinical and clinical drug development.
What it takes to successfully spin out a business from academia
Avance Clinical and Agilex Biolabs share an origin story, formed when CPR Pharma Services split into two in 2019. CPR Pharma Services itself was a South Australian startup, spun out in 2009 from a pharmacokinetics analytical lab at the University of South Australia (UniSA). Jason observed the spinout as laboratory manager at UniSA, and joined Yvonne along with Andrew Dinan as senior managers in CPR Pharma Services.
“One of the most vital factors in spinning out from the university was having support and advocacy from key leaders at UniSA,” Jason explains. “We also worked carefully through some human resource challenges, like being attentive to the details of transitioning people out of academia and into a commercial environment.”
Winning a federal innovation grant along with matched capital investment was also critical.
“That money made it really hard for the university to say no,” says Jason.
For Yvonne, being part of a strong leadership team was key in the early days.
“Part of the reason we were so successful was because we were really passionate, and we had really high regard for each other,” she says. “That allowed us to build a collaborative and supportive environment from the start, which is so critical.”
Building a culture that attracts the right people
Both Avance Clinical and Agilex Biolabs have grown rapidly since 2019. A key aspect of that growth has involved recruiting and retaining staff members who are motivated and committed.
“You’ve got to have a vision of where you’re headed as a business, and then bring people along the journey with you,” Jason says. “And workplace culture is critical – when the culture is right, people want to be there.”
For Jason, one of the core aspects of being a good leader is knowing what your strengths and weaknesses are, and helping your staff attain similar insights.
“Nobody’s perfect at everything and every leader is different,” Jason says. “We do a lot of work to help people understand themselves, including offering opportunities in leadership skills development and mentoring programs.”
Yvonne agrees – and says building a positive workplace culture occurs when high quality, high performance, integrity and a team environment are prioritized.
“We’re working in a highly regulated environment, and we live and die by our quality,” she says. “And because quality and culture are so intertwined, it is absolutely crucial to build an attractive workforce culture in order to attract the best people.”
“This is why we invest in our people, offer flexible work options, and create dedicated office spaces where our teams can collaborate,” says Yvonne.
Understanding the motivations of your board and investors
An effective working relationship between company leaders and board members is a core part of success in the biomedical sector.
“It’s critical to ensure board members understand the business,” says Jason. “And as a leader, you need to understand what the motivations of board members are too.”
Yvonne agrees, thinking back over her experiences working with different kinds of boards – some risk averse, some primarily governance-focused and others more strategic. She says it’s vital for leaders to have awareness of the ambitions of their board, including an understanding of the commercial timeframes investors and board members are operating under.
“But in the end, our organisation is a people-driven business, and so we balance the need to be results-oriented with making sure we really look after our people,” Yvonne says.
South Australia is a top destiny for pre-clinical and clinical research
A number of key factors attract drug development clients to Australia, and South Australia in particular.
“For clients, time is money and they want to minimize their cash burn,” Jason explains. “And so the speed at which we can start things up here is critical.”
“The research and development tax incentive gives companies the opportunity to take some tax breaks as well,” he adds. “And our global best practise standards are clear.”
Yvonne says South Australia has a unique offering for conducting clinical trials.
“In South Australia, we offer integrated solutions for clients to do their clinical research,” she says. “If a client starts here in South Australia, it’s basically like a one-stop shop, because as a state we can offer bioanalytical services, clinical trials services and trial sites, plus we’ve got access to great physicians who are invested in supporting clinical trials.”
“So yes, I’m a strong advocate for South Australia, but also for Australia,” Yvonne says. “Avance Clinical is a global company now with a presence in Australia, New Zealand, the United States and India, and our clients are located all around the world.”